First double-blind vs. placebo clinical study on humans
AlgoSource voluntarily joined an ambitious process aimed at obtaining authorisation from the European Food Safety Authority (EFSA) to make a health-related claim demonstrating, according to the standards of double-blind clinical trials set by the EFSA, the antioxidants properties of Spirulysat®.
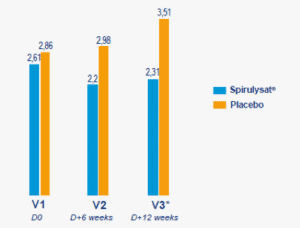
From the end of 2017, AlgoSource launched, with Biofortis Mérieux, the first exploratory clinical study, conducted double-blind against placebo, according to the most stringent clinical standards, the results of which confirmed the functional power of Spirulysat®.
In so doing, AlgoSource undertook the first clinical study on humans using a Spirulina extract, and the only one known to date. So, once again, the company has demonstrated its role as a pioneer in drawing on the wealth of microalgae.
![]()
The second step on the long road to obtaining a health claim consists of conducting a second set of clinical trials, confirmatory, always in double blind, on the basis of a larger number of volunteers. In accordance with the rules of procedure set by the EFSA, this study must be done by a CRO (Contract Research Organization) different from the organization that conducted the exploratory study.
AlgoSource therefore issued a call for tenders to select this second partner who will conduct this second set of clinical trials.
Particularly innovative in the world of food supplements for prevention and supportive care, this approach to obtaining a health claim validated by the EFSA benefits from the support of the Pays de la Loire Region within the framework of the AmiTher project.
Do you have a question?
Contact our team
Do you want to know more about our products, our ingredients from microalgae and our know-how? Contact us!

